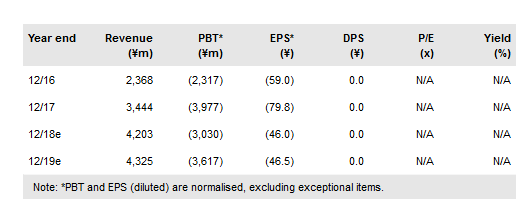
SymBio Pharmaceuticals Ltd (T:4582) announced in October that it has begun preparations to establish its own sales organisation to market Treakisym and other anticancer drugs in Japan after the marketing agreement with Eisai expires in December 2020. The announcement validates our decision earlier this year to adopt self-commercialisation in Japan in our base-case valuation model. Self-commercialisation will improve operating margins and allow it to establish a team of in-house experts to communicate the benefits of Treakisym (and rigosertib if approved) to healthcare providers. Our valuation increases to ¥25.5bn as we roll forward the DCF model; our earnings forecasts and valuation assumptions are unchanged.
Liquid formulations and DLBCL justify own sales
The extension of Treakisym’s lifecycle through the in-license of liquid formulations from Eagle Pharmaceuticals plus the potential for the r/r diffuse large B-cell lymphoma (DLBCL) Phase III to drive further sales growth from 2021 (if successful) support the rationale for SymBio to establish its own salesforce to market Treakisym and other drugs. Treakisym in-market sales in 2017 grew by ~62% to ¥6.8bn in calendar year 2017, supported by two new indications approved in 2016. We model Treakisym sales of ¥10.7bn in FY21, allowing SymBio to attain profitability in the first year of self-commercialisation.
To read the entire report Please click on the pdf File Below..