SymBio Pharmaceuticals Ltd (T:4582) recently obtained approvals for Treakisym in Japan in the additional indications of first-line low grade non-Hodgkin’s lymphoma (NHL) and mantle cell lymphoma (MCL). This follows on from the Aug 2016 approval in chronic lymphocytic leukaemia. We believe these additional indications could nearly double current Treakisym sales (on track for c $40m in 2016) via partner Eisai, which to date have been mostly in relapsed or refractory (r/r) low grade NHL/MCL. These approvals represent an important step in SymBio’s strategy to maximise Treakisym’s potential.
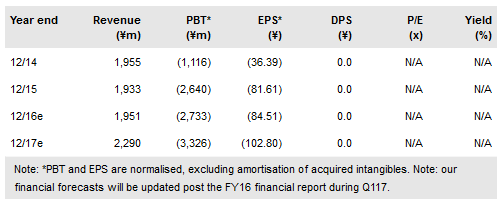
To date, Treakisym sales in Japan have largely been in the c 4,700 patients with r/r low grade NHL/MCL, with around a 60% share, according to SymBio. With c 7,100 first-line low grade NHL/MCL patients and more treatment cycles per patient on average, this indication could materially expand Treakisym’s potential. We expect a formal launch by partner Eisai during Q117. We estimate that SymBio earns an average net margin of 10-12% on top-line Treakisym sales. We believe the main risk to future Treakisym sales will be drug pricing dynamics in Japan, with an increasing focus on cancer. To date, Treakisym pricing has been stable and our forecasts assume continued stable pricing. The next price review will be in 2018.
To read the entire report Please click on the pdf File Below