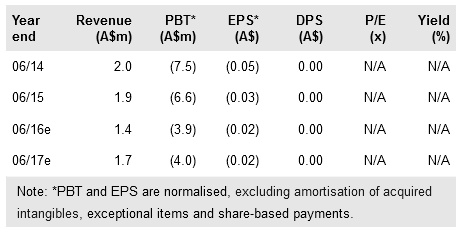
Regeneus Ltd (AX:RGS) reported that its STEP Phase I trial of Progenza in patients with knee osteoarthritis has completed recruitment and that a review of safety data did not identify any safety concerns. The study will complete in H117 when all patients have completed 12 months follow-up. We believe the positive safety review augurs well for the company’s plan to secure a manufacturing and commercial partner for its Progenza mesenchymal stem cell therapy technology in Japan in the current quarter, which may prompt a re-rating of the stock. We leave our valuation unchanged at A$106m (A$0.51/share) ahead of this potential re-rating catalyst.
Positive safety outcome clears way for Japan deal
Progenza is Regeneus’ lead product and contributes more than 60% of our valuation of the company. The Phase I STEP trial of the allogeneic stem cell treatment in patients with knee osteoarthritis has completed recruitment, and a review of cumulative safety data by the study safety oversight committee did not identify any safety concerns. Regeneus is in advanced discussions with a large Japanese company regarding licensing Progenza for the Japanese market. Guidance in the half-yearly report in February was for a deal to be signed by mid-year, and we would expect the clean safety outcome to help move the discussions forward. Directors expect to receive a significant upfront licensing fee on entering this agreement, which is also expected to include further milestone payments.
To read the entire report Please click on the pdf File Below