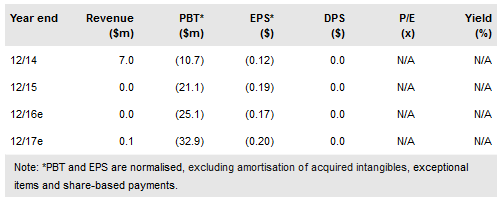
On 27 December the company completed a successful $38m fund-raising and announced a co-promotion deal in the US, which may turn Redhill (NASDAQ:RDHL) into a commercial-stage, revenue-generating gastrointestinal specialty pharma company. In addition, the company revealed final results from the Phase IIa trial for RHB-104 in multiple sclerosis, which echoed positive interim findings earlier in 2016. Next steps will depend on further detailed analysis. Our post fund-raise valuation is NIS1.5bn ($378m) ahead of several key late-stage catalysts in 2017.
Co-promotion deal in the US with focus on GI
A recent major announcement was for a co-promotion deal between RedHill and Concordia Healthcare Corp (NASDAQ:CXRX), according to which RedHill will gain certain rights to promote a gastrointestinal specialty drug Donnatal (phenobarbital, hyoscyamine sulfate, atropine sulfate, scopolamine hydrobromide) in selected regions in the US. Donnatal is a proprietary combination of established compounds, which has anticholinergic (slows down the motility of intestinal muscles) and barbiturate (mild sedation) effects and has been classified by the FDA as possibly effective in irritable bowel syndrome and acute enterocolitis. Donnatal was acquired by Concordia in 2014 and had sales of $71m in H116 (7.7% of Concordia’s sales).
To read the entire report Please click on the pdf File Below