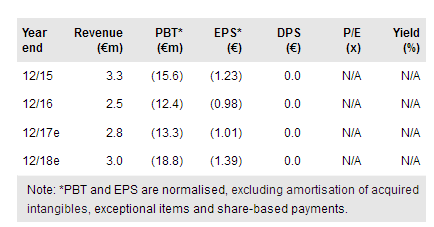
Pixium Vision (PA:PIX) recently reported data from its 10-patient Iris II study showing that it improves visual performance, but stops operating as intended at approximately nine to 12 months post-implantation. Pixium has halted new implantations as it plans to study a potential remedy to improve longevity. We have lowered our success probability estimates and delayed our launch dates for both Iris II and Prima. Our rNPV is reduced to €63.0m, from €129.3m previously.
Six-month data and possible cause of short lifespan
Six-month data shows that Iris II can improve square localisation and direction of motion performance among implanted patients. Pixium believes that mechanical forces (from eye movement) led to microfractures (or breaks) of the wiring emanating from the 150 electrodes embedded on the Iris II device, in the studied patients. There may also be moisture accumulation that caused electrical shorts.
To read the entire report Please click on the pdf File Below: