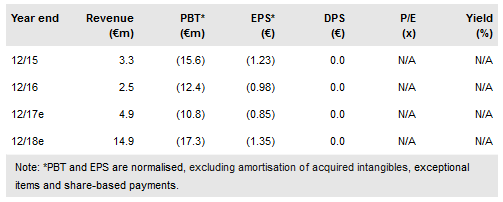
Pixium Vision (PA:PIX) is developing two different retinal implants that provide vision by electrically stimulating the retina. Commercialisation efforts for Iris II are starting in Europe. Prima intends to target a larger population, and human trials could start in H117. Using a risk-adjusted NPV model, we obtain a pipeline rNPV of €131.4m, up from €125.5m, previously.
With CE Mark, Iris II gearing for initial EU sales
The Iris II epi-retinal implant received CE Mark clearance in 2016, and is designed to partially restore vision in profoundly blind retinitis pigmentosa (RP) patients. Pixium is building up a sales channel across Europe and the Middle East, and finished recruitment for a 10-patient study. It is seeking conditional reimbursement in Germany and France, and in February 2017 received a positive decision in Germany where it was granted NUB Status-1 (full approval). This allows selected hospitals to negotiate with insurance providers to obtain maximum Iris II coverage.
To read the entire report Please click on the pdf File Below