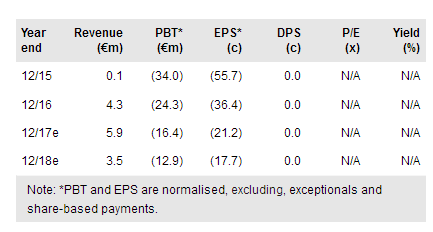
Paion O.N (DE:PA8G) remains on track to file for approval of Remimazolam in 2018 for procedural sedation (via partner Cosmo Pharmaceuticals in the US) and general anaesthesia (GA) in Japan. Remimazolam’s excellent safety profile and shorter procedure times than midazolam are expected to drive uptake if it gains market approval. Preparations are underway for an EU Phase III study for GA in general surgery patients to start in 2018 (subject to funding). Recently granted formulation and dosing patents will strengthen market exclusivity in the EU and Japan until 2033, which sees us lift our valuation to €260m (vs €245m) or €4.25/share.
Abuse potential study last hurdle before US filing
Paion has conducted Phase I studies to assess the pharmacodynamics of intranasal administration of remimazolam and whether it can be abused as a knockout cocktail combined with alcohol. It will hold discussions with the FDA in Q417 ahead of a second intranasal study to be able to comprehensively understand the potential for abuse. Contingent on study results and FDA discussions, Paion expects to complete the human abuse liability programme in H118. Partner Cosmo expects to file for approval in H218.
To read the entire report Please click on the pdf File Below: