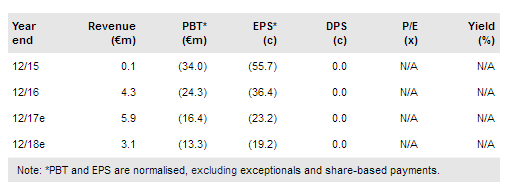
Paion O.N (DE:PA8G) has completed patient recruitment in its pivotal bronchoscopy study of its ultra-fast-acting sedative remimazolam. Results are likely by mid-2017 and, if positive, would put it on track to file for US approval for procedural sedation in mid-2018. Paion is also preparing a dossier for a filing for general anaesthesia (GA) in Japan in a similar time frame. It has also outlined a programme that could see it restart Phase III studies in GA in Europe (estimated cost €20-25m). Paion has sufficient cash to fund operations beyond end 2018, but would need additional funds to complete the Europe development programme. We leave our valuation unchanged at €214m ahead of the key bronchoscopy trial results.
Bronchoscopy data likely in June
On 27 March Paion announced full recruitment in the confirmatory Phase III trial of remimazolam for procedural sedation in bronchoscopy patients, with headline data expected in mid-2017. Based on consultation with the FDA, Paion will initiate additional Phase I studies to further assess abuse potential. A pre-NDA meeting is planned for the end of 2017, with a US filing by partner Cosmo expected in mid-2018 (pending positive trial results).
To read the entire report Please click on the pdf File Below