The recent fund-raising, coupled with the Sanofi (SNY) opt-in payment, means Oxford BioMedica (OXB.L) is well funded and permits the development of its flexible LentiVector gene-delivery technology through to several value inflection points. ProSavin, to correct the dopamine depletion of Parkinson’s disease, may be the most advanced project, but it is the earlier-stage ocular programmes that elicit most interest. The promising data means management is increasingly prioritising this area and re-positioning the business accordingly.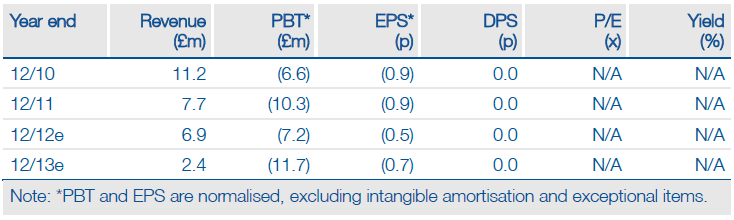
Ocular projects are the main focus
The LentiVector gene-delivery platform is well suited to ophthalmic indications, with a single administration offering the potential of a permanent benefit. There are six projects in various stages of development, with Sanofi collaborating on four programmes targeting specific indications (having recently opted in for StarGen and UshStat). A number of Phase I/II trials are underway, with further data expected to be reported during Q412. RetinoStat, for age-related macular degeneration, has the largest commercial potential and will be the focus of attention.
Manufacturing site now fully approved
The new LentiVector production facility is now fully operational, marking a material de-risking of the whole technology. Direct control of the manufacturing process is seen as a major advantage in partnering discussions, with additional benefits from the cost savings (compared to outsourcing) and the potential to capture more of the value chain. Additionally, management has not ruled out using any spare capacity for third-party manufacture, since the extra income would be welcome, but emphasises that in-house production remains the principal focus.
Valuation: Risk-adjusted NPV of £62.5m
Oxford BioMedica’s investment case rests principally on the successful development of its ocular programmes in collaboration with Sanofi, notably the potential opt-in to development/commercialisation licences for RetinoStat and EncroStat, and on securing partners/funding for ProSavin and TroVax. Our valuation of £62.5m is based on the rNPV model of the R&D pipeline. We employ conservative assumptions and include only late-stage or highly visible assets, leaving upside potential for earlier-stage programmes and the financial benefits arising from the in-house production facility.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Oxford Bio Medica Funded Through To Value Inflection Points
Published 10/10/2012, 08:16 AM
Updated 07/09/2023, 06:31 AM
Oxford Bio Medica Funded Through To Value Inflection Points
Outlook improving...
Latest comments
Loading next article…
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.