Orexo AB (ST:ORX) appeal of the US District Court’s invalidation of the ‘330 patent, which had been anticipated for some months, has been successful. Zubsolv is Orexo’s largest and fastest-growing product – we anticipate sales of c SEK620m and over 25% growth in FY18. The initial invalidity of the ‘330 patent had weighed on the shares since 2014 and this overhang has now been removed. The exclusivity of the Zubsolv patents runs until 2019 and 2032. We have updated our valuation ahead of the Q318 results.
Orexo wins its appeal of the District Court’s invalidation of the Zubsolv ‘330 patent
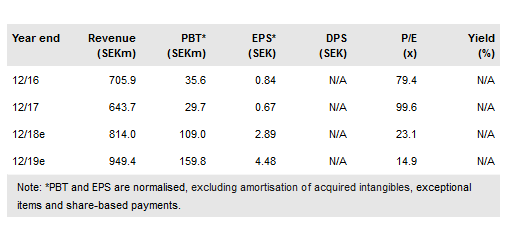
We have updated our model for our sales expectations for the Q318 results and a number of other changes that now include Zubsolv US exclusivity until 2032. Orexo has done an exceptional job in growing branded Zubsolv revenues in the face of competition from a genericised opioid-dependency category. Our experience in the pricing and reimbursement space suggests that Orexo’s successful US market access and reimbursement strategy for Zubsolv was eventually bound to attract a serious intellectual property challenge in an attempt to replicate the product’s success.
To read the entire report Please click on the pdf File Below: