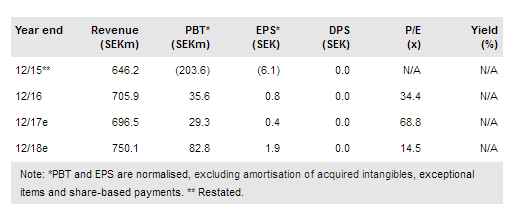
Orexo (ST:ORX) has received a $2.5m (c SEK22m) milestone from AstraZeneca on the start of Phase I trials for respiratory programme, OX-CLI (also known as AZD9898). Orexo now has two clinical assets (the other being Phase III-ready acute pain programme OX-51) and three preclinical projects (OX-MPI [inflammation] plus two novel formulation technologies). This milestone does not impact FY17 guidance but indicates growing momentum in the early-stage development pipeline. We expect further news on new product opportunities this year; both pipeline project(s) and commercial product(s) for US promotion, in addition to potential EMA approval of Zubsolv (Q417).
A growing pipeline beyond Zubsolv
Of Orexo’s five R&D stage assets OX-CLI is the only one partnered. Licensed to AstraZeneca in 2016, Orexo is eligible for success-based development/commercial milestones and a tiered single-digit net sales royalty. Partnering discussions are ongoing for OX-MPI and OX-51, but deal timing is unknown. Orexo is also applying its significant formulation technology expertise to generate an early-stage pipeline: two novel oral and second-generation sublingual formulations are being assessed. Subject to demonstrating proof of principle, more disclosure is expected in H217.
To read the entire report Please click on the pdf File Below