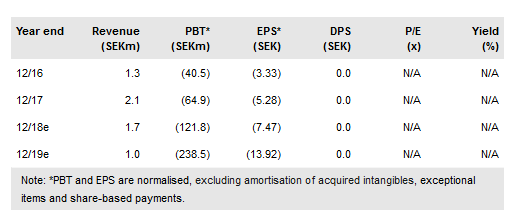
Oncology Venture (TE:ONVE) attended the annual American Society of Clinical Oncology (ASCO) in June with three abstracts. Results from a Phase I study of 2X-121, a dual PARP-1/2 and TNKS-1/2 inhibitor, as a single agent in patients with solid tumours were presented along with data on the development of the 2X-121 drug response predictor (DRP) algorithm. OV also provided supplementary data supporting the LiPlaCis and APO010 programmes at the conference. In late May, OV announced the merger between OV and the Medical Prognosis Institute (MPI) was approved by the board of directors.
2X-121 data detailed in ASCO presentation
OV presented data from the 41-patient Phase I study of 2X-121as a single agent in patients with advanced solid tumours, along with data on the development and preliminary testing of the 414-gene 2X-121 DRP algorithm in 13 patients. The responders and non-responders were identified with median overall survival of more than 800 days and 208 days, respectively. The company expects to initiate the focused Phase II trial this year.
To read the entire report Please click on the pdf File Below: