Sharper focus on anti-cancer drugs
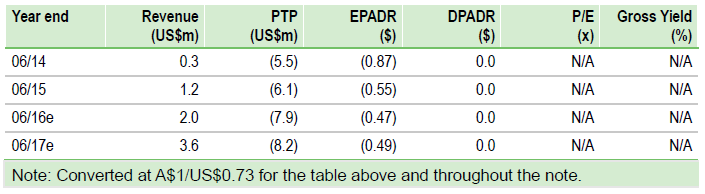
Novogen (O:NVGN) has prioritised development of its three lead anti-cancer drugs following a scientific review. Cantrixil is scheduled to enter the clinic in H116 and Anisina in H216 (both pending successful completion of toxicology studies). We have trimmed our valuation model, removing the prostate cancer indication for Trilexium, as it is not expected to enter the clinic until 2017, which reduces our valuation to $87m (vs $126m) or $5.13 per ADR (undiluted). With $32m cash, Novogen is funded beyond FY18.
Scientific review leads to sharper focus
Following a review of its drug development program with the assistance of a scientific advisory board, Novogen has sharpened its focus on the clinical development of Cantrixil and Anisina ahead of Trilexium. Initial development of Cantrixil is likely to focus on ovarian cancer rather than a broader malignant ascites indication. For Anisina, prostate cancer continues to be seen as a promising adult cancer indication, although the company is evaluating other cancer indications in preclinical studies. The company reaffirmed its commitment to a paediatric neuroblastoma program for Anisina. Trilexium is now not expected to enter the clinic until H216/H117 and brain cancer and melanoma indications are being prioritised over prostate cancer as adult indications. The brain cancer Diffuse Intrinsic Pontine Glioma continues to be assessed as a paediatric cancer indication.
To Read the Entire Report Please Click on the pdf File Below