Newron (SIX:NWRN) and its partners Zambon and U.S. WorldMeds have announced that the FDA has approved Xadago (safinamide) for Parkinson’s disease (PD) patients as an add-on therapy to levodopa/carbidopa.
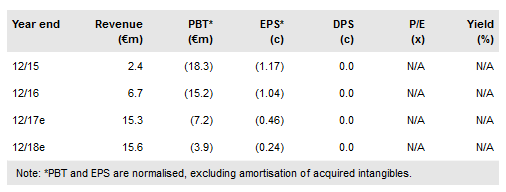
While we had expected approval (following the revised PDUFA date of 21 March), the certainty of approval removes a major overhang on the stock, and as such we expect a positive share price reaction. We expect launch in the U.S. market in H217 by sub licensee U.S. WorldMeds. Our valuation of Newron remains unchanged at CHF530m.
Commercial partner Zambon has sub licensed its U.S. commercialisation rights to U.S. WorldMeds and we expect U.S. launch in H217; the U.S. approval will trigger a €9m milestone payment to Newron. U.S. WorldMeds is a Kentucky-based pharmaceutical company that markets a number of specialty products including a treatment for PD (APOKYN for the acute treatment of intermittent ‘off’ episodes). Xadago is a neat fit into its portfolio given its indication for use as add on to levodopa/carbidopa in PD. U.S. WorldMeds plans to focus more than 60 sales representatives on launching Xadago in the U.S.
To read the entire report Please click on the pdf file below: