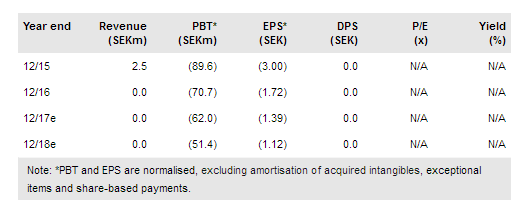
NeuroVive Pharmaceuticals AB (ST:NVP) is a mitochondrial medicine specialist with a diversified asset portfolio. It employs a two-pronged strategy and has a portfolio of drug candidates for orphan mitochondrial diseases, which it aims to develop internally; more recently, it has also identified in-house assets suitable to tackle larger indications, which NeuroVive aims to out-license in pre-clinical development. The most advanced projects are Phase IIb ready NeuroSTAT for traumatic brain injury and KL1333 in Phase I for various mitochondrial diseases. We value NeuroVive at SEK1.5bn.
Core portfolio focuses on orphan diseases
NeuroVive’s core portfolio, which the company aims to develop internally, targets orphan indications: traumatic brain injury (TBI) with NeuroSTAT, various genetic mitochondrial diseases with KL1333 and NVP015, and mitochondrial myopathy with NVP025. Recent R&D news regarding the positive outcome in a Phase IIa study with NeuroSTAT (novel ciclosporin A formulation) means that the drug candidate is ready for a proof-of-concept study. The second most advanced product KL1333 (NAD+ modulator) was in-licensed from Yungjin Pharm in May 2017 and currently is in Phase I.
To read the entire report Please click on the pdf File Below: