Mologen develops anti-cancer immune maintenance therapies aiming to give long- lasting responses. The investment case rests on a possible 2013 deal on the lead project, MGN1703, based on high-quality interim Phase II data. These indicated a 50% reduction in the hazard risk of disease progression when MGN1703 was dosed every three days. Mologen aims to partner MGN1703 to fund internal development of MGN1601, a cell-based vaccine for metastatic renal cancer. MGN1601 could be an orphan drug with no generic version possible; Mologen intends to sell direct.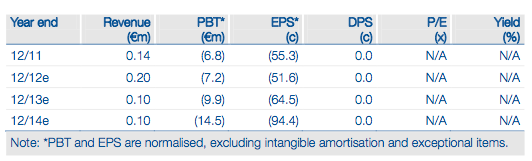
MGN1703 -- A Clear Colorectal Cancer Maintenance Effect
MGN1703 is a stable, dumbbell-shaped DNA molecule (dSLIM) that activates innate immunity. Interim data in metastatic colorectal cancer (mCRC) showed that the hazard ratio for disease progression was reduced by 50% compared to a matched placebo group on an intent-to-treat (ITT) basis. Statistical significance was seen when a group of nine ineligible and high-risk patients were excluded. Mologen aims to partner MGN1703 in 2013, but will progress into Phase IIb/III independently of a deal.
MGN1601 -- Cell-Based Renal Cancer Vaccine Plus dSLIM
This allogenic renal cancer cell vaccine plus dSLIM has reported data for a 19-patient Phase I metastatic renal cancer safety study, ASET. Immune effects were detected in some patients and one partial response was seen with some stable disease cases. There are good data for a small, initial study. Mologen is developing a Phase II design in metastatic renal cancer patients. If MGN1601 is approved, Mologen plans to sell direct in the EU and US. Generic competition will not be possible as this is a unique, proprietary cell-line, so MGN1601 could yield valuable long-term profits.
Valuation: MGN1703 Deal Value Of €18.20, Un-Partnered €16.50
The indicative value of Mologen depends on whether a deal on MGN1703 occurs either in 2013 or after Phase III data in 2016. A deal in 2013 would fund Phase II development of MGN1601. However, if a good deal is not feasible, Mologen can progress both studies, but would need a further €25m with a better deal possible in 2016, depending on Phase III data. MGN1703 may face generic competition in the EU in 2025 and in the US in 2023. A major value assumption is that MGN1601 is free from direct generic competition, as it uses a proprietary cancer cell line. It is also assumed that Mologen can sell this orphan product direct to maximise long-term profits. Edison has valued Mologen with a 2013 MGN1703 deal at €18.40 per share, assuming 45% MGN1703 and 25% MGN1601 probabilities. A regulatory-stage 2016 deal indicates about €16.50 per share value.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Mologen: The Investment Case
Published 02/25/2013, 11:01 AM
Updated 07/09/2023, 06:31 AM
Mologen: The Investment Case
Innately Adaptive
Latest comments
Loading next article…
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.