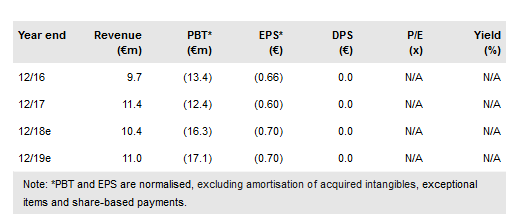
Medigene NA O.N. (DE:MDG1k) MDG1011 trial in MM, AML and MDS is ongoing and the first MDG1011 TCR cell product has been successfully produced. Additionally, procedures to speed up patient enrolment are being rolled out, including the simplification of enrolment criteria and the addition of new trial centres. We continue to forecast that clinical data from both the Phase I part of the MDG1011 Phase I/II clinical TCR trial and the now fully enrolled Phase I/II dendritic cell (DC) vaccine trial will be available in 2019. Financials for 9M18 were above guidance, driven by a reduction in expected R&D costs. We now forecast a net loss of €16.9m in FY18 vs €18.4m previously. Additionally, Medigene announced an exclusive licence agreement with Leiden University to develop a TCR against HA-1, an antigen expressed in a range of cancers. We value Medigene at €457m.
MDG1011 TCR trial: Patient screening ongoing
The first personalised MDG1011 TCR therapy has been manufactured in the Phase I/II clinical trial and met the requirements needed for patient administration. However, due to the progression of the patient’s disease, the treatment could not to be administered. Screening of patients is ongoing, although strict enrolment criteria (eg PRAME+ and HLA-A*02:01+) mean that only 10–20% of potential patients are eligible for treatment. We forecast initial Phase I data in 2019.
To read the entire report Please click on the pdf File Below..