Becoming an increasingly prominent player
Medigene NA O.N. (DE:MDG1k) is well funded (FY16 cash €52.6m) to advance both its DC vaccine programmes and TCR programme. We expect a number of important milestones in 2017; specifically, we expect newsflow from its most advanced technology (DC vaccines) in Phase I/II studies for AML (complete enrolment) and the start of its first company-initiated T-cell receptor (TCR) clinical study. We have increased our rNPV-based valuation to €293m (vs €233m), to reflect the increase in the TCR programme probability to 13% (vs 5%), rolling the model forward and using FY16 cash.
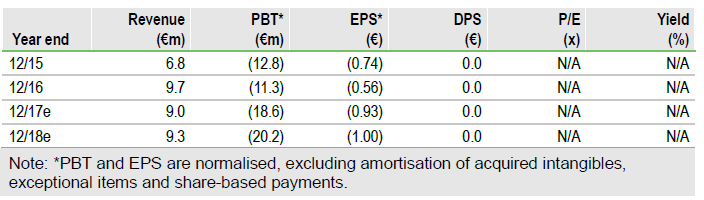
FY16 results demonstrated good progress
Medigene made good progress in 2016 by realising value from a number of its non-core assets (sale of its Catherex subsidiary, AAVLP deal, final transference of EndoTAG-1 to SynCore and sale of 50% of its stake in Immunocore shares), alongside expansion of its TCR technology platform with the grant of a US patent. Underpinning this was the development of its senior management team, which Medigene believes is now in place to move the company forward. Of particular significance was the announcement of its TCR development deal with bluebird bio, a prominent T-cell immunology company. This was Medigene’s first commercial partnering agreement based on its proprietary TCR technology platform and is important as it validates Medigene’s technology and offers potentially new immuno-oncology products, while retaining all rights for its TCR programme and pipeline.
To read the entire report Please click on the pdf File Below