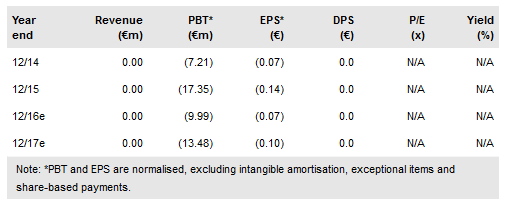
Kiadis Pharma BV (AS:KDS) has presented one-year follow-up data from a Phase II study in leukaemia patients (n=23) at the 58th meeting of the American Society of Hematology (ASH) in San Diego, US. Patients were administered ATIR101 after a haematopoietic stem cell transplant (HSCT) from a partially matching donor. The primary endpoint of transplant-related mortality (TRM) and secondary endpoint overall survival (OS) for patients receiving HSCT+ATIR101 were significantly higher than patients receiving HSCT alone from an observational control group. We are encouraged to see the low incidence of relapse in the ATIR101 arm in this high-risk patient population. Our valuation is €383.2m or €27.4 per share.
ATIR101: Increasingly positive data at one year
In this open-label single dose Phase II study, ATIR101 administered after HSCT showed a significant reduction in the primary endpoint of TRM at 32% vs 70% for HSCT only (p=0.007) of a historical comparator group. Overall survival was 61% for HSCT+ATIR101 vs 20% for HSCT only (p=0.0023). There were no cases of grade III-IV acute Graft vs Host Disease (GVHD), only three cases of grade II acute GVHD, and one chronic GVHD. Thus, event-free survival rate (GRFS) was 57% after one year, which is higher than 20% for the historic group, even for matching unrelated donors (41%) and post-transplant cyclophosphamide (PTCy) of 33% as reported by Solh et al. (2016).
To read the entire report Please click on the pdf File Below