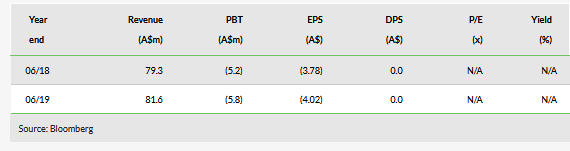
Ellex Medical Lasers (ASX:ELX)'s products target diseases associated with growing ageing populations such as glaucoma and age-related macular degeneration (AMD). FY19 revenue was up 3% to A$81.6m, as robust growth in iTrack, SLT lasers and 2RT was offset by declines in laser devices for other indications and in diagnostic equipment. iTrack, SLT lasers and 2RT appear well-positioned to continue their growth trajectory in FY20 and there may be some recovery in Ellex’s remaining product lines.
SLT laser and iTrack showing growth in glaucoma
Ellex’s SLT laser effectively lowers intraocular pressure (IOP), and SLT laser revenue grew 10% in FY19 to A$32.4m. Sales could further benefit from the H219 publication in Lancet of the LiGHT study (n=718), which compared SLT to hypotensive eye drop use in treatment-naïve patients, and found that at three years, SLT-treated patients were within their target IOP at 93% of visits vs 91.3% in the eye drops arm. The study stated that SLT should be offered as a first-line treatment (and not only for those failing eye drop therapy). Ellex also reported a 29% sales increase (to A$14.3m) of its iTrack MIGS system, positioned for cases where further IOP control is needed, with unit volumes up 17% (30% in US). Compared to other MIGS devices such as Glaukos’s iStent, the iTrack ABiC procedure does not need to be performed in conjunction with cataract surgery and does not involve permanent device implantation. SLT and iTrack should benefit from favourable demographics and increasing adoption of non-topical treatment methods.
2RT provides a huge opportunity in iAMD
AMD is a leading cause of vision loss among persons over age 55 and Ellex’s 2RT intends to treat intermediate AMD (iAMD) by applying a nanosecond nonthermal pulse laser targeting selected macular retinal pigment epithelium (RPE) cells to promote extracellular repair. 36-month data from the LEAD study (n=292) reported in 2018 showed that in patients without coexistent baseline reticular pseudodrusen (RPD), 2RT treatment led to a 77% reduction in the rate of progression. This data sparked interest in 2RT, as FY19 sales rose 260% to A$1.8m. Recently reported 48-month data revealed similar trends, and Ellex expects to establish a regulatory pathway (including a new study) with the FDA by YE19. If US approval is eventually received, 2RT could potentially become Ellex’s best-selling product given the wide unmet need for iAMD treatment (as no treatment has been approved by the FDA).
Valuation: EV of A$80m
Ellex FY19 net loss was A$5.8m, and it expects a lower loss in FY20 as it seeks to contain costs. Ellex had A$0.3m net cash at 30 June 2019 (gross debt of $15.1m vs $15.4m gross cash), although most of its debt is due within 12 months.
Share price graph
Historical financials
Business description
Based in Australia, Ellex Medical Lasers markets laser surgical and medical devices for the ophthalmic sector. Its SLT and iTrack Ab-Interno Canaloplasty (ABiC) devices aim to treat glaucoma, and 2RT is being developed for intermediate AMD, an area of large unmet need as no current treatment exists.