XF-73, Destiny Pharma PLC (LON:DEST)’s new antimicrobial, is in Phase IIb for the prevention of staphylococcal surgical infections and seems to be in the right place at the right time. Recent press reports suggest that in most developed markets, cutting screening for nasal staphylococcal carriage before surgery was associated with patient deaths. Another recent paper highlighted the mortality benefit of nasal decolonisation.
Reduced screening in hospitals has negative effects
A recent press report from Scotland has linked the deaths of five hospitalised patients to reductions in the programme of screening for methicillin-resistant Staphylococcus aureus (MRSA) to prevent hospital-acquired infections. Screening fell from 92% to 60% of hospitalised patients. This is not surprising as preventative measures involving medical staff at admission, laboratory analysis and a clinical decision are convoluted and easy targets for cost cutting although MRSA bacteraemia surveillance has been mandatory in the UK since 2001. We believe any reduction in screening, although difficult to condone, is positive for Destiny’s XF-73 product for nasal decolonisation because its rapid bactericidal activity and lack of resistance could support the use in most patients admitted to hospital, without screening (saving costs) and should reduce post-surgical deaths.
Recent NEJM paper mirrors Destiny’s strategy
A recent paper in the New England Journal of Medicine compared the effect of nasal decolonisation on MRSA infection versus hygiene education alone. The nasal decolonisation regimen included off-label nasal mupirocin, which has its limitations with bacterial resistance and reimbursement. These issues are not relevant to Destiny’s XF-73 nasal formulation. Nasal decolonisation was associated with a 30% lower risk of MRSA infection over education alone. The number of patients needed to be treated (NNT) to avoid one infection was only 30. The NNT figure is more supportive of Destiny’s XF-73 nasal formulation, which has a shorter dosing period than nasal mupirocin’s three times a day for five days. In addition, XF-73’s shorter on-label preventative indication should ease reimbursement.
Valuation: Unchanged
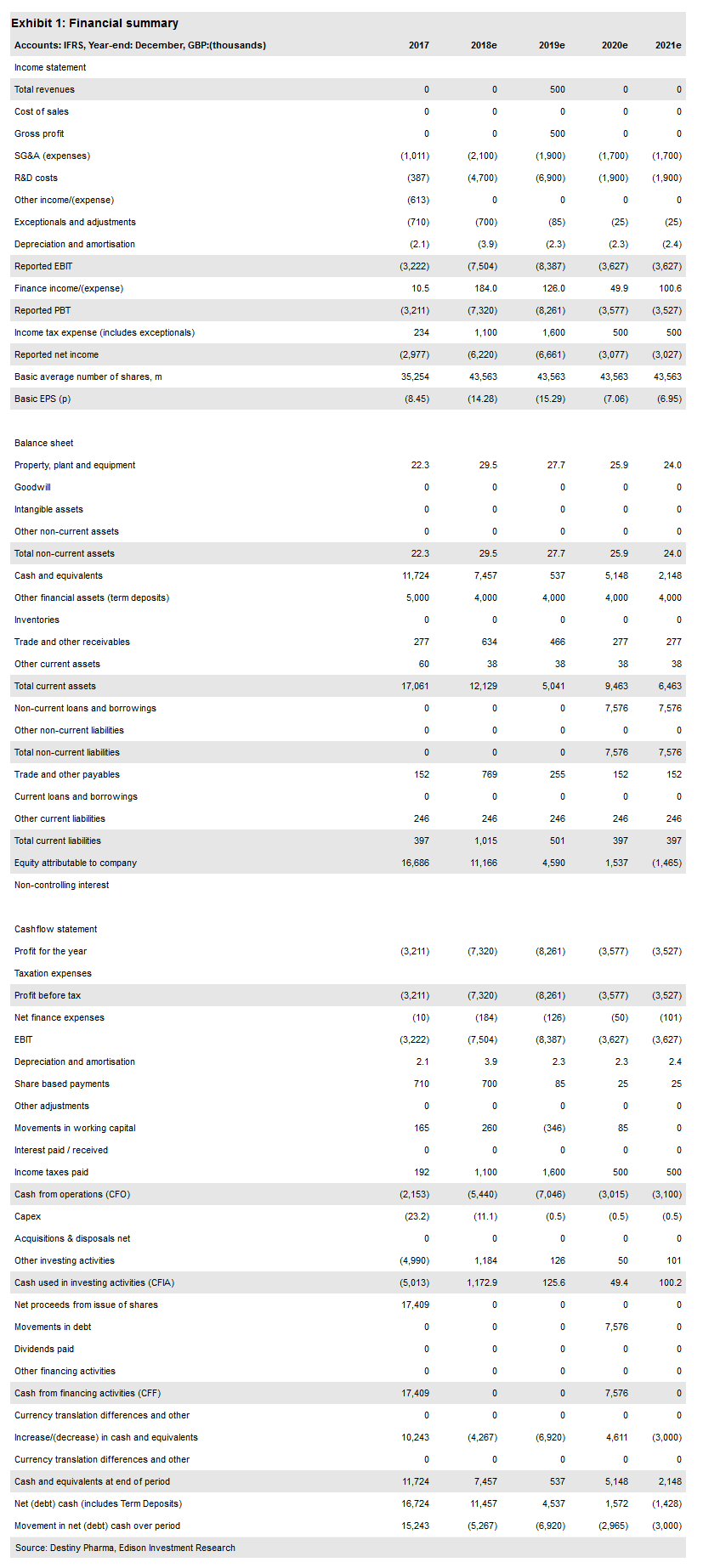
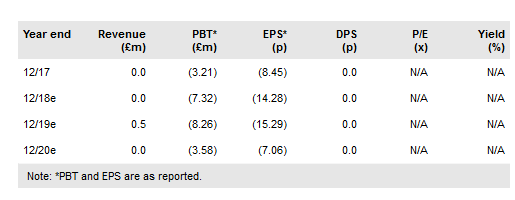
We have not changed our valuation ahead of Destiny’s FY18 results in April. With the supportive commentary on the prevention of post-surgical infections, we may also revisit our assumptions on the market penetration of XF-73. At present, our model values XF-73 use only in high-risk screened surgical patients (neurosurgery, some cardiovascular and orthopaedic procedures). Our valuation remains £86.7m or £1.99 per share.