Destiny Pharma PLC (LON:DEST) has had two positive and supportive announcements. The U.K. government has announced a number of initiatives and strategies to significantly enhance the return on investment for companies like Destiny that are developing new drugs to combat antimicrobial resistance (AMR). In addition, Destiny has been awarded a £1.6m U.K.-China AMR grant.
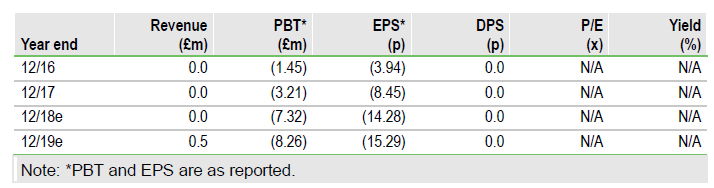
The U.K. government has released a five-year action plan and a 20-year vision to tackle AMR, which follows similar proposals from the FDA in June 2018. The U.K. proposals include financial incentives and additional investment for companies such as Destiny Pharma that are developing new antimicrobial drugs. Our initiation report noted that Destiny’s lead product, XF-73, is active against Gram-positive and some Gram-negative bacteria that include all strains of methicillin-resistant Staphylococcus aureus tested. This puts Destiny on the front line in the fight against AMR and it should benefit from these initiatives.
The U.K.’s new five-year AMR national action plan includes ‘stimulating the development of new products’ active against AMR bacteria. This is in addition to Lord O’Neil’s 2016 AMR review, where the pay-or-play concept of a government imposed antibiotic investment charge on the pharmaceutical sector but not on companies that invest in AMR drug development, was suggested. This thinking has been extended with the U.K. government’s recent strategies targeting the low returns on investment on AMR drug development by a new assessment of the cost-effectiveness of antimicrobial agents and new payment models. Because antimicrobial agents such as XF-73 typically cure patients, cost-effectiveness should be obvious.
Destiny has also announced a grant of up to £1.6m from the U.K.-China AMR grant fund set up by Innovate U.K. on the eradication of resistant bacteria by its XF-series drugs. Innovate U.K. administers a number of peer-reviewed grant award schemes.
Business Description
Destiny Pharma is dedicated to the discovery, development and commercialization of new antimicrobial agents that have unique properties to improve outcomes for patients. Destiny’s first product, XF-73, is about to start a US Phase IIb clinical study.