BiondVax Pharmaceuticals Ltd’s (NASDAQ:BVXV) FY16 business update reiterated that the company and its partners are on track to deliver full results from the ongoing Phase IIb trial with multimeric vaccine candidate M-001 by end Q217. According to plans, the US National Institutes of Health (NIH) will initiate the last Phase II study with M-001 in 2017, which will pave the way for partnering and the Phase III programme. Currently BiondVax is a leader in the development of a universal influenza vaccine. Our valuation is increased to NIS278m ($77m); on a per share basis it is down slightly to NIS1.7/share ($18.2/ADR) after the recent fund-raising.
Financials: Keeping the operations lean
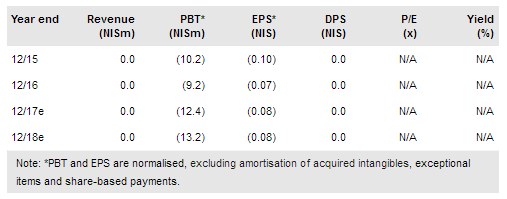
BiondVax has reported end-FY16 cash and cash equivalents (cash, cash equivalents and short- and long-term marketable securities) of NIS27.4m ($7.1m), before announcing a private placement of net NIS10.9m ($2.8m) in January 2017. FY16 R&D expenditures were NIS7.8m ($2.0m), in line with our expected NIS7.9m ($2.1m), while MG&A costs were somewhat higher than our estimate of NIS3.4m ($0.9m) but, in our opinion, still reasonable at NIS4.1m ($1.1m). According to our model, cash reach is beyond 2019 assuming a similar level of activities. Therefore, Phase III studies are achievable with current cash, but will require additional funding which, according to BiondVax, could come from non-dilutive sources.
To read the entire report Please click on the pdf File Below