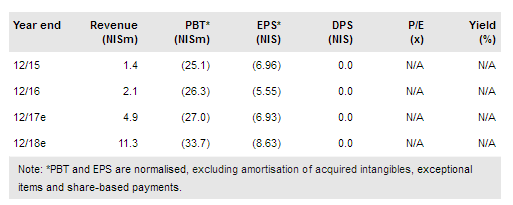
BioLight Life Sciences Ltd (TA:BOLT) reported positive results from a 77-patient US Phase I/IIa study on Eye-D VS-101, being developed as an extended-dose platform for treating glaucoma. We have raised our estimate for the candidate’s probability of commercial success to 30% (from 20%), which, with other adjustments, leads to an increase in our rNPV valuation to NIS121.6-135.7m.
24% reduction in Diurnal IOP at 12 weeks
The Eye-D VS-101 is an insert that is placed in the lower lid conjunctiva in an in-office procedure that delivers a controlled amount of latanoprost. In the Phase I/IIa study, patients were randomised into four groups, with three receiving a VS-101 insert (at differing dose levels per group) and the control group receiving latanoprost 0.005% eye drops. BioLight has not yet specified the precise dosing quantities for the VS-101 treatment arms, but indicated that one of the doses showed a 24% reduction in diurnal (daytime average) intraocular pressure (IOP) from baseline (from 23.5mmHg to 17.9mmHg). The firm reported that the VS-101 insert was well-tolerated, and most adverse events were expected, and found to be mild and transient, although we await further details in a subsequent publication or conference presentation.
To read the entire report Please click on the pdf File Below: