Basilea Pharmaceutica AG (SIX:BSLN) reported a solid set of 2016 numbers ahead of guidance, driven by higher than expected product sales in H216 combined with lower than expected operating costs. Cresemba (for invasive mould infections), in particular, continues to pick up momentum both in the EU and the US through US partner Astellas.
Zevtera’s US pivotal Phase III clinical trial programme for bacterial infections remains on track to initiate in mid-2017. Multiple distribution and commercialisation agreements signed in H216 should lead to international launches starting from 2018 for both products. We value Basilea at CHF1,048m or CHF97/share.
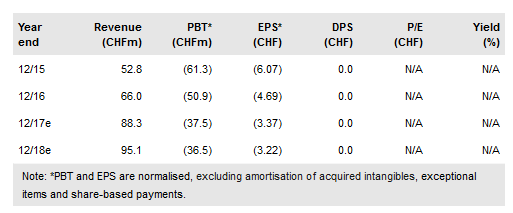
FY16 results: Guidance beaten; still room to grow
Basilea reported total revenues of CHF66m in FY16 (+25%), including product sales of CHF7.1m (relating to the first full year in which both brands have been available in Europe), which beat guidance of CHF5m. This was due to Cresemba uptake in the EU, although Zevtera uptake remains lacklustre (we have lowered our peak sales expectations accordingly).
Basilea reported CHF7.3m in royalties on $46m of US Cresemba sales reported by Astellas. 2017 guidance for product sales and royalties (on US sales) stands at CHF15m and CHF14m respectively. In 2018 both products should benefit from international launches following the slew of distribution deals in H216; we anticipate further deals in 2017.
To read the entire report Please click on the pdf File Below