Basilea Pharmaceutica (SIX:BSLN) has made a solid start to the first half of 2016, with anti-microbial agents Cresemba and Zevtera continuing their respective European roll-outs. Ongoing dialogue with the FDA on the SPA protocol for Zevtera’s Phase III US clinical trial program means US trials should initiate in H117.
Together, these underpin the current valuation with upside potential from further partnering deals for Cresemba, Zevtera and the early-stage oncology pipeline, which in itself is making progress, albeit at an early stage. We value Basilea at CHF1,091m or CHF101/share.
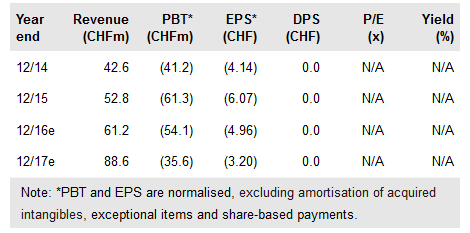
Cresemba and Zevtera underpin current share price
Basilea has confirmed its 2016 outlook following the interim results. We anticipate a CHF5m sales contribution for FY16 from Cresemba for mould infections and Zevtera for bacterial infections against a backdrop of an ongoing European roll-out for both.
Basilea has a contract salesforce in place (via Quintiles) to commercialize these in the core European markets, with product overlap likely to drive future operational synergies. Partnering activities, particularly for Zevtera in the US, could provide upside.
To read the entire report, please click on the PDF file below: