Astex (ASTX) has expanded its Phase II trial of SGI-110 to 200 patients, by adding a new cohort of relapsed/refractory (r/r) myelodysplastic syndromes (MDS) patients to the existing front-line acute myeloid leukemia (AML), MDS and r/r AML groups. Enrolment in the study is now over half complete and data from the r/r AML cohort should be available for presentation at the American Society of Hematology (ASH) meeting in December. We have reviewed our valuation in the light of separately reported preclinical data on SGI-110 in ovarian cancer, and now value Astex at $678m (including cash), equivalent to $7.25 per share basic or $6.24 per share (fully diluted).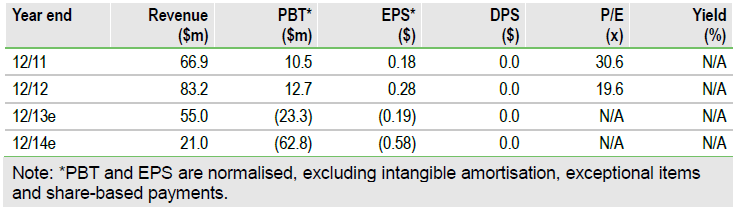
Progress of SGI-110 is now driving share value
SGI-110, Astex’s second-generation hypomethylating agent (HMA), is receiving renewed investor interest because of its proven mechanism of action and potential advantages over first-generation HMAs. Astex expects to determine its registration strategy for the product on completion of the ongoing Phase II trial in MDS/AML. Assuming a Phase III start in a haematological indication in 2014, SGI-110 could reach the market by 2016. With the global HMA market exceeding $1bn per year, SGI-110 could generate a significant revenue stream for Astex.
Data on SGI-110 in ovarian cancer boosts valuation
Despite an early clinical study being underway, ovarian cancer had previously not been included as supportive data had been generated with Dacogen, rather than SGI-110, albeit showing an impressive overall response rate (ORR) (35%) in platinum-resistant or -refractory patients. The recent publication of data from preclinical studies of SGI-110 in ovarian cancer now justifies adding this indication in our valuation model. SGI-110 is in a Phase I/II trial in a group that could produce proof-of-concept data in 2014. It is also in an exploratory Phase I/II trial in hepatocellular carcinoma and this indication is not yet included in our valuation.
Valuation: Upside remains
The addition of a contribution for SGI-110 in ovarian cancer adds $80m to our valuation, which rises to $678m (including cash), equivalent to $7.25 per share basic or $6.24 per share (fully diluted). We contend that our approach remains conservative, principally valuing only Dacogen, SGI-110 (in haematology and ovarian cancer) and AT13387 (in ALK+ NSCLC), with a modest contribution from Astex’s partnered compounds. We see significant potential value increases as SGI-110 and AT13387 advance into later stages of development in the next 12 to 18 months.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Astex Pharmaceuticals: Progress of SGI-110 Is Now Driving Share Value
Published 04/16/2013, 07:38 AM
Updated 07/09/2023, 06:31 AM
Astex Pharmaceuticals: Progress of SGI-110 Is Now Driving Share Value
SGI-110 moving along
3rd party Ad. Not an offer or recommendation by Investing.com. See disclosure here or
remove ads
.
Latest comments
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.