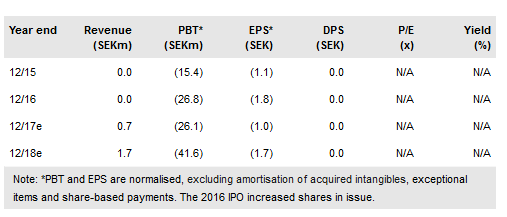
Acarix A/S (ST:ACARIX) has laid the groundwork in 2017 for sales development in 2018. The main subset of the key Dan-NICAD study was published in November 2017 and should boost medical awareness. As initial sales were slower than expected, sales forecasts have been adjusted although, with only two quarters’ sales data, these will be revised regularly. Good sales growth in Europe is expected from 2019. With no US trial announced our expected launch date is moved to 2022 from 2021. The indicative value is now SEK448m (formerly SEK728m), implying a fair value of SEK19.46/share (formerly SEK31.62/share). A new CEO will be appointed in 2018.
CAD-score – a clear indicator of low coronary risk
The Acarix diagnostic device uses a highly sensitive microphone linked to a minimalist, self-contained processing module to record a patient’s diastolic heart sounds. There is a single-use patch to attach the device to the patient’s chest. The CADScor V3 algorithm (validated by the Dan-NICAD clinical study) calculates a CAD-score rating. A negative CADScor result was correct in 96% of tested patients, according to Winther et al (2017). The new Dan-NICAD II study will add more validated CAD patients to further improve the innovative scoring algorithm to gain further acceptance of this new risk scoring method among key opinion leaders.
To read the entire report Please click on the pdf File Below: