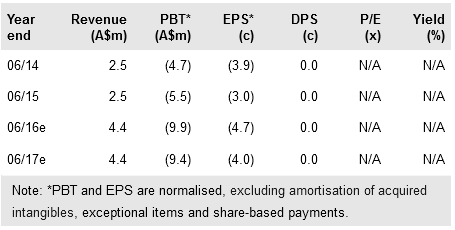
Impressive initial results from the MITCI Phase Ib trial suggest that Viralytics Ltd (AX:VLA) could push to the forefront of the crowded field of immune checkpoint inhibitor (ICI) combination therapies. The first six melanoma patients treated with Cavatak in combination with Yervoy (ipilimumab) experienced a higher response rate (67%) and lower serious adverse event rate (9%) than other prominent ICI combination therapies. These encouraging initial data bode well for the ongoing Phase Ib trials of Cavatak in combination with Merck’s Keytruda in melanoma, lung and bladder cancer, and are likely to be of great interest to potential partners. We increase our valuation to A$272m (A$192m previously), or A$1.15 per share (was A$1.04).
Impressive preliminary response rate in MITCI
Preliminary results from the ongoing MITCI Phase Ib trial of Cavatak in combination with Yervoy (ipilimumab) in patients with advanced melanoma were presented at AACR in April. Of the 11 patients treated to date only one (9%) has experienced a serious (grade 3 or higher) adverse event. Four of the six (67%) patients who have been assessed for tumour response have experienced confirmed objective responses (including two complete responses), compared to response rates of 28% and 11% reported for Cavatak and Yervoy, respectively, as single agents and response rates of 56% and 60% for Yervoy in combination with Imlygic (T-vec) and Opdivo. Separately, an update on the CAPRA trial of intra-tumoural injection of Cavatak in combination with the anti-PD1 ICI Keytruda is expected in H216.
To read the entire report Please click on the pdf File Below