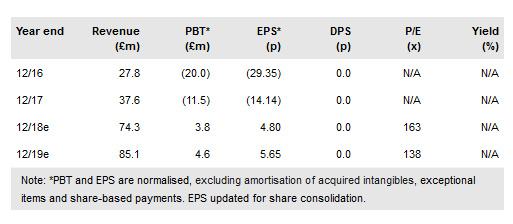
Strong operational momentum at Oxford BioMedica PLC (LON:OXB), as evidenced by its interim maiden operating profit of £9.4m (vs a loss of £2.2m in H117), highlights the strength in the company’s diversified business model. We continue to expect ongoing growth in the top line, driven in the near term by the commercial ramp-up of Kymriah (Novartis), the progression of Bioverativ’s haemophilia products to the clinic and the rapid advancement of its partnered products with Orchard and Axovant. We note that Immune Design’s CMB305 clinical programme has been halted, but forecast that the operational and financial impact on OXB will be minimal. OXB has transitioned three new preclinical assets into its pipeline: OXB-204 (Ophthalmology-LCA10), OXB-208 (Ophthalmology- RP1) and OXB-103 (amyotrophic lateral sclerosis). Additionally, OXB has announced the expansion of its lentiviral manufacturing capacity with a fourth facility. We value OXB at £632m vs £614m previously.
Growth the focus, profit the benefit
Growth in H118 platform (£25.0m) and product revenues (£10.2m) was aided by new partnerships/deals with Bioverativ (£8.1m) and Axovant (£10.2m) respectively. Remaining platform revenue of £15.4m (H117: £13.1m) was predominately driven by bioprocessing/commercial development revenue from the Novartis and Orchard collaborations. Despite increases in H118 R&D to £14.1m (H117: £10.5m), COGS of £10.1m (H117: £8.0m) and financial costs of £4.2m (H117: £3.7m), net income for H118 was a £5.1m profit. We forecast a FY18 net profit of £3.1m (FY17:-£9.0m).
To read the entire report Please click on the pdf File Below..