Personalized immunotherapy for MS
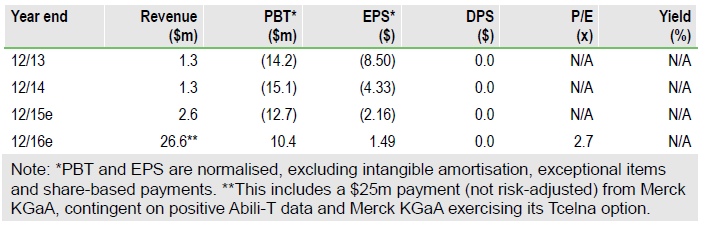
Opexa’s Tcelna immunotherapy is advancing in Phase IIb studies in secondary progressive MS (SPMS), with data expected in H216. The firm is fully funded to Phase II data which, if positive, could sharply increase investor and stakeholder interest. Merck KGaA (N:MRK) has an option to in-license Tcelna in MS. Opexa (O:OPXA) is also developing OPX-212 in neuromyelitis optica (NMO), a rare autoimmune disorder leading to vision loss and paralysis. Opexa plans to file an IND and start a Phase I/II study in H116. Our rNPVderived equity valuation is $51m.
ImmPath aims to suppress autoimmune responses
Opexa’s core platform, ImmPath, produces patient-specific (autologous) T-cell immunotherapy following the collection of a patient’s own blood. The blood cells are screened against predefined potential self-reacting (autoimmune) target proteins, and the dominant self-reacting T-cell lines are isolated and expanded. An attenuated end-product is reinjected into the patient, which aims to suppress these undesired self-reacting T-cells and thus curb autoimmune disease progression.
To Read the Entire Report Please Click on the pdf File Below