Neovacs (PA:ALNEV)'s lead project, IFN-Kinoid (IFN-K) for lupus, started a 178-patient EU, US and RoW Phase II in Q315. It expects data by mid-2017. This is based on clinical response and measurement of the interferon signature (IS), a diagnostic marker of lupus.
CKD, a leading Korean pharmaceutical company, partnered the product for Korea in 2015; marketing may start in 2018. Neovacs plans to partner IFN-K, implying possible launches in 2021. VEGF-Kinoid for cancer and AMD could start Phase I in H117 and a type 1 diabetes kinoid is in preclinical testing. Cash in December 2015 was €6.1m; a French rights issue in June 2016 raised €8m gross at €0.85/share.
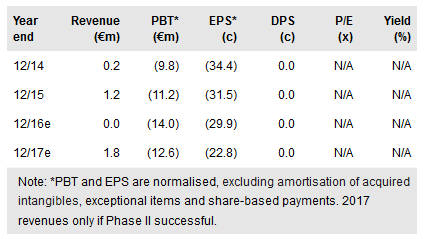
Phase IIb data due June 2017 then partnering
Neovacs is aiming to complete recruitment into the 178-patient Phase IIb by late summer 2016, with an expected June 2017 release of preliminary data on the two combined, 36-week endpoints. Neovacs has FDA approval to include US centres.
The trial is based on the neutralisation of the interferon gene expression signature, now seen as a key lupus marker. It will also score clinical responses assessed using the BILAG system.
Two IFN-based candidates from AstraZeneca have shown that patients with high interferon signatures respond better to IFN control. Neovacs intends to find a partner to run and fund Phase III studies from 2018. This could enable a US and EU regulatory filing by 2020 and possible marketing from 2021.
To read the entire report Please click on the pdf File Below