Starting the new year with a bang
GW Pharmaceuticals, (GWP) has raised $101.1m gross through a follow-on offering of 2.81m new ADSs at $36.00/ADS. These funds will be used to accelerate Epidiolex development in orphan childhood epilepsies (Dravet syndrome and Lennox-Gastaut syndrome), while importantly allowing GW to retain global commercial rights. Phase I epilepsy data and the start of Phase II trials of Epidiolex are expected during 2014. Overall, 2014 remains potentially transformative for GW as its pipeline delivers important clinical milestones. Major inflection points for lead product Sativex include Phase III cancer pain data (Q414) and US Phase III start in MS spasticity (H214).
$101.1m (gross) raised on Nasdaq
GW’s oversubscribed secondary offering of 2.44m new ADSs (representing c 29.3m new ordinary shares) at $36 each closed on 14 January; the 15% overallotment option (366k ADSs or c 4.4m ordinary shares) was also exercised. Net proceeds were $94m (£57.1m).
Advancing Epidiolex development for epilepsy
GW has secured funding for Phase II and III development of Epidiolex (cannabidiol, CBD) in refractory childhood epilepsy, and for Epidiolex supply to US physicians under Expanded Access treatment Investigational New Drug Applications (INDs). To date, the FDA has approved seven physician-led INDs for use of Epidiolex in c 125 children with refractory epilepsy; initial data is expected from mid-2014. Following conclusion of FDA discussions regarding the Phase II programme design during H114, GW expects to initiate a Phase II trial of Epidiolex in Dravet syndrome (FDA orphan status has been granted) in H214. Proceeds will also contribute to the GWP42006 Phase II in adult epilepsy, early-stage pipeline progression (including glioma), building a US staff presence and expansion of Epidiolex manufacturing.
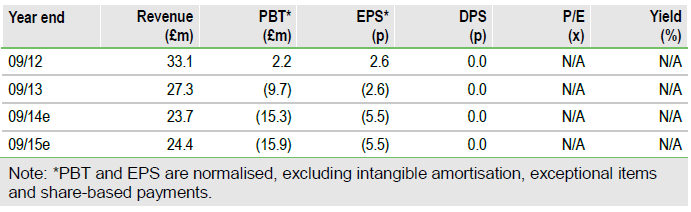
Valuation: Fair value of £692m, with near-term upside
Our updated model reflects the fund-raise, new cost guidance, future Sativex sales by Ipsen and potential of Epidiolex in Lennox-Gastaut syndrome. This increases our risk-adjusted DCF to £692m (previously £447m), or 320p. Positive data from physician-led Epidiolex studies and Phase II start in Dravet syndrome (mid-2014 onwards) would increase our valuation to £772m (357p/share). Positive Sativex Phase III cancer pain data (late 2014) would raise our DCF further to £803m (371p/share). GW’s remaining R&D programmes (epilepsy, ulcerative colitis and schizophrenia) currently offer pure upside to our valuation.
To Read the Entire Report Please Click on the pdf File Below