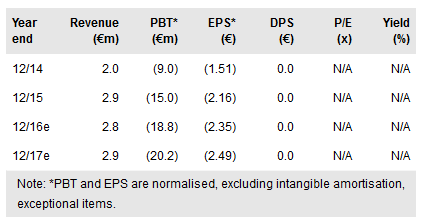
Erytech Pharma (PA:ERYP) lead indication, acute lymphocytic leukaemia (ALL), is being explored in a comprehensive programme for its Graspa technology, with acute myeloid leukemia (AML) and pancreatic cancer following next. Key clinical data are expected for all three indications during the next 12 months, with the hallmark event being expected approval of the marketing application for Graspa for relapsed/refractory ALL in Europe by end-2016/early 2017. Our valuation of Erytech remains largely unchanged at €322m.
Key catalysts to mid-2017
With Graspa already filed in Europe in relapsed/refractory ALL and a decision expected by end-2016, the focus is now on the US and new indications. A Phase Ib study in ALL in the US is ongoing with data expected in 2016. This could potentially allow rolling out development into front-line ALL globally. A Phase IIb trial in AML is more than 90% recruited, with primary results expected in mid-2017. A Phase II trial in pancreatic cancer is the first solid tumour indication and results should be released in Q416.
To read the entire report Please click on the pdf File Below