EpiCept’s investment case is centred on finding a development partner for AmiKet, a product for chemotherapy-induced peripheral neuropathy. Although EpiCept’s valuation is depressed as a result of the ongoing regulatory and commercial setbacks with respect to Ceplene and Azixa, the upside from a successful deal -- that might include an M&A transaction -- could be considerable. Cash currently lasts to Q3, while our valuation -- presuming Amiket is partnered -- is a multiple of the current share price.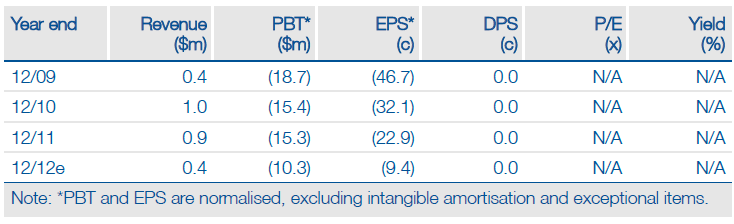
AmiKet Granted Fast-Track Designation By FDA
The U.S. FDA’s recent award of Fast Track designation to Amiket for chemotherapy-induced peripheral neuropathy (CIPN) should shorten regulatory timelines and could help attract a development partner. In January, EpiCept received finalised FDA guidance for the Phase III clinical development plan. EpiCept also recently received EMA guidance that is consistent with the FDA, which should make it easier to design a Phase III programme for filing an NDA in both the U.S. and EU that is faster and cheaper.
F573 Emerges From Preclinical Studies For Viral Hepatitis
F573 (EP1013), a small molecule licensed to GNI Group for Asia, Australia and New Zealand, has demonstrated a therapeutic effect in a preclinical acute liver injury model, including a reduction in TNF-alpha and cell apoptosis. An Investigational New Drug (IND) application was filed in China by GNI in July 2011. EpiCept retains ex-Asia rights to the compound.
Financials: Funded To Q312
An April 2012 $1.1m ($0.97m net) fund-raising of convertible preferred stock (convertible into 6.3m ordinary shares) should extend the cash runway into Q312. EpiCept ended Q112 with $5.1m of cash.
Valuation: Risk-Adjusted NPV Of $79m
EpiCept has a current market cap of $12.6m, a senior secured loan of $6.6m and cash of $5.1m as of 31 March 2012 resulting in an EV of $14.1m. In comparison, we calculate a risk-adjusted NPV of $79m based on prudent assumptions of the four products’ probability of success in each indication, launch date, pricing and market penetration.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
EpiCept's AmiKet Gets FDA Fast Track
Published 06/13/2012, 03:05 PM
Updated 07/09/2023, 06:31 AM
EpiCept's AmiKet Gets FDA Fast Track
Moving Forward With Pain
Latest comments
Loading next article…
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.