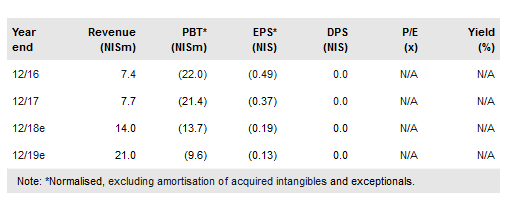
Allium Medical Solutions Ltd (TA:ALMD) has reported H118 financial results. Revenues increased 31% y-o-y to NIS4.9m. As before, we continue to expect additional product approvals during 2018 and sales growth from its distribution deals in new and established markets. We maintain our FY18 sales forecast of NIS14m with a projected 2017-2020e revenue CAGR of 57%. We continue to believe that regional expansion is crucial to the company’s investment case. Our valuation, updated for net cash of NIS16.6m, is NIS1.82 per share.
H118 revenues 31% higher; FY18 forecast maintained
Revenues in H118 were up 31% to NIS4.9m vs NIS3.7m in H117. Sales have been driven mainly by Allium Stents in Europe and approvals in new territories, mainly China. Other marketed products are EndoFast fixation device for urogynecology in Europe and Israel, and Gardia’s embolic protection system in Europe and the US. Importantly, the gross margin increased to 40% from 25% in H117 due to economies of scale, higher prices and higher efficiency of the production process. R&D spend was similar to H117 (NIS7.3m and NIS7.1m respectively), while S&M expenses were up 33% due to increased marketing activity. G&A expenses grew 37% to NIS4.9m due to non-cash costs associated with stock-based compensation resulting from an option grant worth NIS769k and one-off costs for Allevetix patents. We maintain our FY18 revenue forecast of NIS14m as we expect sales to accelerate in H218 due to increasing marketing activity in new territories. We also maintain our FY18 EBITDA loss forecast of NIS13.2m (NIS11m loss in H118) as some H118 expenses were one-off. That said, we see some downside risk to our estimates. We expect cash of NIS16.6m at end H118 to provide runway into H219.
To read the entire report Please click on the pdf File Below: