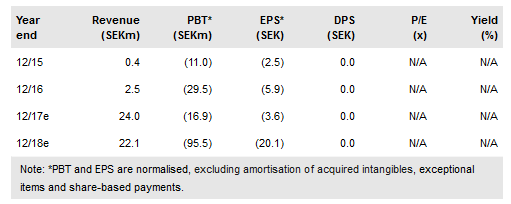
With Italian GMP approval of the Spherotide one-month formulation, Xbrane Biopharma AB (ST:XBRANE) can ship a SEK7m order to Iran. A Chinese deal worth SEK17m upfront, $8m total, may be signed in H117. European partnering and launches are possible from 2019 after clinical trials. Xbrane (or a partner) may sell Xlucane, its low-cost biosimilar of Lucentis (2015 sales $3.6bn), in the US after 2021 and from 2022 in Europe. A study has been done on pilot scale batches showing high biosimilarity, giving a solid basis for scale up.
Prostate cancer: GMP status for triptorelin generic
Triptorelin, sold as various brands, is one of a well-known class used to treat advanced prostate cancer, endometriosis and uterine fibroids. Sales in 2015 for these indications were $380m. Initial Spherotide sales are planned in 2017 with a SEK7m order to Iran which can now be shipped as Xbrane has good Manufacturing Practice (GMP) certification from the Italian authorities. An agreed, but not signed, Chinese $8m deal could yield SEK17m cash in H117, with sales perhaps from 2021/22. EU marketing is expected from 2019 via a partner and a further deal.
To read the entire report Please click on the pdf File Below