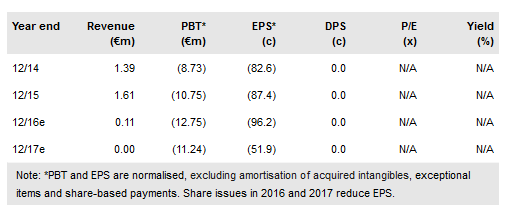
Txcell's (PA:TXCL) novel CAR-modified regulatory T-cell (CAR Treg) platform is developing well. CAR Tregs offer a powerful and versatile new approach to autoimmune and immune system disorders. TxCell has four indications in preclinical development with the first ever CAR Treg trial, in transplant rejection, anticipated by TxCell to start by late 2018. This could provide powerful clinical proof-of-concept data by 2020. Ovasave for Crohn’s disease remains as a clinical-stage project but is currently on hold. TxCell used a convertible loan facility in 2016 drawing €4.9m in cash. The indicative market cap is now €74m and focussed onto CAR Treg.
Focus on CAR Treg with four preclinical projects
TxCell is developing two platform technologies: ENTrIA and ASTrIA. The ENTrIA platform uses chimeric antigen receptor (CAR) technology similar to that in the CAR T-cell cancer area. We expect ENTrIA to be an excellent basis for partnering and technology licensing. A granted European patent (licensed globally in June 2016) offers broad protection. CAR Treg trials may start from 2018 with transplant, based on a key academic collaboration. Other indications cited are lupus nephritis, bullous pemphigoid (skin) and multiple sclerosis. Ovasave, using non-modified Tr1 Tregs, is on hold while manufacturing is validated.
To read the entire report Please click on the pdf File Below