Transgene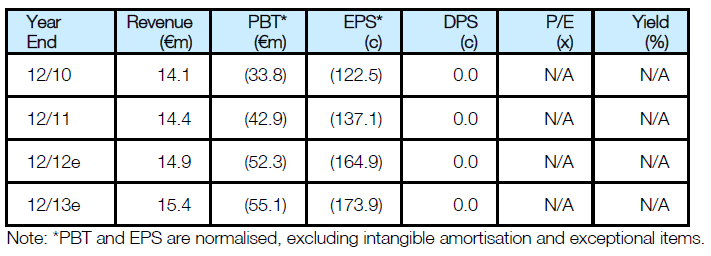
Investment Summary: A Setback But That Is All
Transgene’s shares have fallen by over 25% since the setback with TG4001, but we have only reduced our valuation by 9%. This product was the least valuable of Transgene’s four-Phase II products and still has potential in HPV-associated cancers. The more valuable Phase-II programmes are progressing as expected. There was more promising data on TG4040 in HCV, posters on TG4010 and JX-594 will be presented at ASCO in June and recruitment for the Phase-II/III TIME trial with TG4010 is progressing.
TG4001 Efficacious, But Not Enough For CIN
Data from the Phase-IIb trial (n=206) with TG4001 in CIN (cervical intraepithelial neoplasia, pre-cancerous cervical lesions) showed the TG4001 increased histological resolution to 25% compared to 10% with placebo at six months. However, for TG4001 to have a commercial future it was considered that a 60% resolution rate was required. So, TG4001 is no longer being developed for CIN.
Potential In HPV-Associated cancers
TG4001, probably with chemotherapy, still has potential to become a treatment for head and neck and cervical cancer. In the CIN Phase-IIa trial, viral clearance was 37% in patients receiving TG4001 compared to 14% on placebo. HPV infection causes cervical cancer and is increasingly the cause of head and neck cancer. Development in these new indications depends on a partner being found.
TG4040 Continues To Show Promise In HCV
24-week data from the Phase-II trial in HCV with TG4040 continues to show the product’s potential. The next step is a combination study with a new direct-acting antiviral agent, such as Gilead’s GS7977 or Bristol-Myers Squibb’s daclatsavir (both in Phase-III development). It is expected start in 2012 and report data in 2013.
Valuation: DCF Reduced By €52m To €554m
Our valuation of Transgene is lowered to €554m after the setback with TG4001, compared to the current market cap of €233m. The next catalysts for the shares are the four posters that will be presented at the ASCO meeting at the beginning of June; one on the Phase-II/III TIME trial with TG4010 and the others with interim analyses and information on JX594.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Transgene Update
Latest comments
Loading next article…
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.