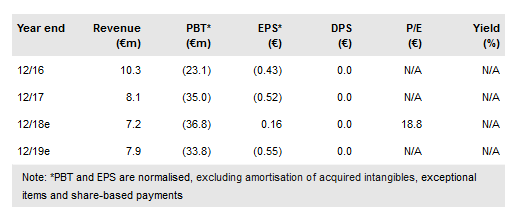
Late-stage clinical trial readouts in 2019 will be critical to Transgene (PA:TRNG) immunoncology (IO) aspirations and, if positive, could further its position in the sector; notably efficacy data expected from the Phase II TG4010 (+nivolumab +chemotherapy) trial in first line non-small cell lung cancer (NSCLC) and the Phase III Pexa-Vec (+sorafenib) trial in first line hepatocellular carcinoma (HCC) (trial conducted by partner SillaJen). Next-generation platforms Invir.IO and myvac continue to progress, with assets from both expected to enter the clinic in 2019. Additional financing is needed to ensure a cash reach beyond September 2019. We value Transgene at €4.68 per share (€290m).
TG4010: First-line NSCLC efficacy data in H219
Transgene’s strategy involves developing its immunotherapies for the treatment of cancer and viral indications in combination with other products, predominantly immune checkpoint inhibitors (ICIs). Following the completion/termination of five trials and the addition of the Phase I TG6002 trial in gastrointestinal adenocarcinoma, Transgene now has six ongoing studies across five products. Efficacy data from the Phase II TG4010 (+nivolumab +chemotherapy) trial in first-line NSCLC be central to determining Transgene’s long-term IO strategy.
To read the entire report Please click on the pdf File Below..