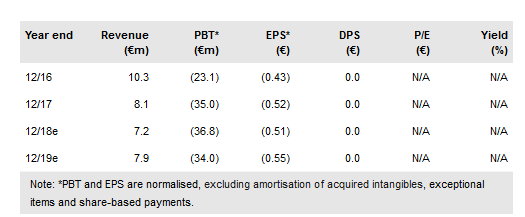
Transgene (PA:TRNG) develops virus-based product candidates for use in oncology and infectious diseases, its five clinical products are currently in 11 clinical trials across a variety of indications, its most advanced is Phase III trial with Pexa-Vec in first-line Hepatocellular Carcinoma with data anticipated in 2019. Its strategy remains to develop its candidates in combination with approved treatments, notably immune checkpoint inhibitors (ICIs). It anticipates eight clinical readouts across its five clinical candidates by year end, notably TG4010 in combination with Opdivo and chemo in first-line NSCLC. We value Transgene at €4.65/share vs €3.80/share previously.
All five product candidates to readout by year end
By year end Transgene forecast ICI combination readouts for TG4010 in first and second line NSCLC, TG4001 in head and neck cancer, in addition to Pexa-Vec in both first line hepatocellular carcinoma and solid tumours. This breath of data will give initial indications of whether Transgene’s ICI combination strategy is valid; future funding and development opportunities will be significantly influenced by these readouts. Outside of ICI combination data, readouts for TG1050 in hepatitis B, TG6002 in glioblastoma and Pexa-Vec in breast cancer are anticipated in H218.
To read the entire report Please click on the pdf File Below: