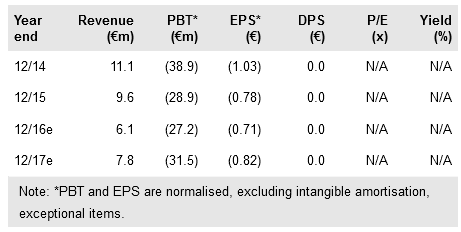
Transgene (PA:TRNG) has successfully completed a major restructuring programme and secured fresh financing (up to €30m from the EIB and Institut Mérieux), which puts it in a strong position to advance its promising immunotherapy technologies once more. The focus is on conducting new Phase I/II studies for Pexa-Vec (oncolytic virus) and TG4010 (MUC1 cancer vaccine) in combination with immune checkpoint inhibitors, an eagerly anticipated approach for hard-to-treat cancers. Partner SillaJen has started a Phase III trial with Pexa-Vec for liver cancer. We re-initiate coverage of Transgene with a base-case valuation of €160m or €4.15 per share.
Positive progression of lead candidates
The strategy of combining novel agents with immune checkpoint inhibitors (ICIs), to help increase the number of patient responders to ICIs beyond the current 20-40%, is a widely accepted concept that holds significant potential. Transgene plans to start Phase I/II combination trials of Pexa-Vec and TG4010 with ICIs (nivolumab + ipilimumab) in 2016, and positive data from these studies would significantly enhance fresh partnering and/or financing options.
To read the entire report Please click on the pdf File Below