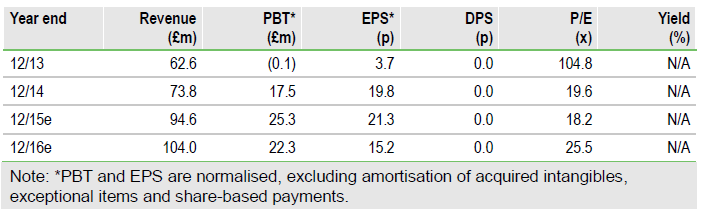
A shining start to 2016
Skyepharma (L:SKP) ended 2015 with a flurry of positive news. The partnering deal with Mundipharma for SKP-2076, Pacira’s announcement of a resolution achieved with the FDA on Exparel coupled with an encouraging FY15 preclose update marks a good start for 2016. Skyepharma has entered into a feasibility and option agreement for SKP-2076 with Mundipharma, the partner of choice in our view, and the timely announcement could ensure seamless development once SKP-2076 feasibility studies complete by mid- 2016. Meanwhile, the FDA has formally rescinded a warning letter issued to Pacira on US Exparel marketing and, importantly, also broadened the Exparel label significantly.
Mundipharma a natural choice
Skyepharma has entered into a feasibility and option agreement with Mundipharma for SKP-2076, a potential asthma therapy that combines three established drugs (ICS/LAMA/LABA) into one inhaler. While we had hoped for such a collaboration, its execution enables Skyepharma to focus on the feasibility studies, paving the way for clinical development in 2016 when Mundipharma has the option to fund the full development and commercialisation under a global licence. Our 2016 forecasts now include receipt of a non-refundable option fee that we estimate at £250,000.
To read the entire report Please click on the pdf File Below