With its FY15 results, Probiodrug AG (AS:PDB) provided an update on the lead Phase IIa SAPHIR trial, which is well underway. R&D expenses were up year-on-year reflecting increased activities related to the study. The six-month delay was mainly a result of enrolment difficulties, which is not uncommon in Alzheimer’s disease (AD) studies due to increasing competition for the same early patient population. Importantly, measures taken by the company addressed this issue well, in our view. The final results are likely in late Q117 or early Q217 which, if positive, should enable Probiodrug to seek a partnership deal.
Well-managed G&A costs, funded likely until readout
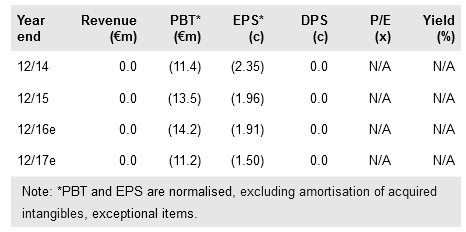
While 2015 R&D costs were up by 27% year-on-year to €10.2m (in line with our expectations) reflecting the initiation of the Phase IIa SAPHIR study, G&A expenses were kept under control and stayed flat. Probiodrug has indicated that the net loss for 2016 may be larger than that incurred in 2015 due to additional costs related to SAPHIR. Based on our updated forecasts we believe Probiodrug is funded until readout following the private placement, which raised €13.5m in November 2015.
To read the entire report Please click on the pdf File Below