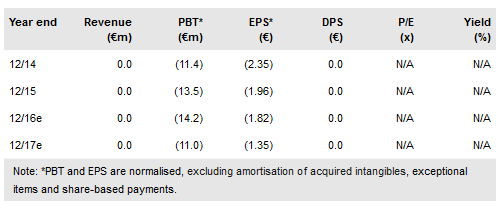
Probiodrug AG (AS:PDB) announced today that the Phase IIa trial with its lead product PQ912 is now fully enrolled with 120 patients surpassing the initial target of 110 and the data will be released in Q217. The trial is investigating the effects of first-in-class PQ912, a small molecule glutaminyl cyclase (QC) inhibitor, on Alzheimer’s disease (AD) patients. Although primarily a safety and tolerability study, a set of exploratory readouts should provide a glimpse of the effect PQ912 has on the pathology of the disease. Following the successful fundraise of €14.9m via the issue of new shares (10% of the existing) in October 2016, our valuation of Probiodrug is €337m or €41.2/share.
Baseline mean MMSE indicates high quality data
Probiodrug mentioned that at baseline the mean Mini-Mental State Examination (MMSE) score of all patients was 25.3 (out maximum of 30) with low variability. Such high score implies a very mild dementia and is well in line with inclusion criteria of MMSE scores being in the range 21-30. Low variability of the scores allows us to expect high quality data and fewer outliers that can skew the findings. The final results should be reported in Q217.
To read the entire report Please click on the pdf File Below