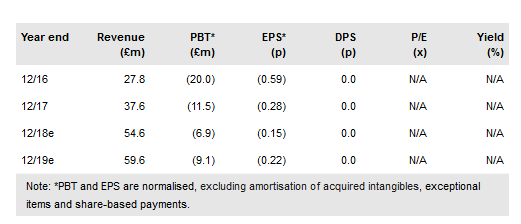
Following the commercial launch in 2017 of partner Novartis’s CAR-T Kymriah (in r/r paediatric ALL), Oxford BioMedica plc (LON:OXB) has become one of a handful of regulatory approved gene and cell therapy manufacturers worldwide. This validation of its capabilities was most recently demonstrated in signing the £100m+ collaboration with Bioverativ to develop haemophilia gene therapies. We expect OXB to rapidly expand its manufacturing capacity to enable top-line growth. We have reassessed many of our valuation assumptions and now include the partnerships with Immune Design, Orchard and Bioverativ. Moreover, internal asset, OXB-102, will start a Phase II trial shortly. We value OXB at £513m (15.6p/share).
Top-line growth the focus as market expands
The global cell and gene therapy market is expanding rapidly as evidenced by the first approvals for both ex vivo (chimeric antigen receptor T-cells [CAR-Ts] Kymriah and Yescarta) and in vivo (Strimvelis for ADA-SCID and Luxturna for RPE65 inherited retinal disease) gene therapy products. As a leading supplier of lentiviral products for gene therapy, OXB is well placed to benefit and recently completed a net £19.3m capital raise to fund the construction of additional manufacturing capacity. We expect the new facilities to be commercially operational by end 2019.
To read the entire report Please click on the pdf File Below: