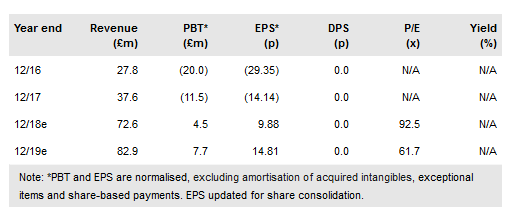
Oxford BioMedica PLC (LON:OXB) has signed an out-licensing deal with Axovant for its Parkinson’s disease (PD) gene therapy AXO-Lenti-PD (previously OXB-102) worth up to $842.5m. Axovant plans to accelerate it rapidly into the clinic, with a Phase I/II dose escalation study in advanced PD patients to be initiated by year end. In addition to OXB’s numerous other partnerships, notably the recently signed Bioverativ deal and the ongoing collaboration with Novartis on its launched CAR-T Kymriah, this deal demonstrates OXB’s continuing world-leading status in lentiviral technology. We now include the Axovant deal in our model and value OXB at £614m.
Commercial revenue opportunity significant
The out-licensing deal for OXB’s AXO-Lenti-PD (previously OXB-102) adds to a growing and diversified revenue base. The deal includes $30m upfront ($5m as a prepayment for manufacturing-related activities), $55m in development milestones and $757.5m in commercial milestones in addition to tiered royalties of 7-10%. AXO-Lenti-PD is a re-engineered version of OXB’s gene therapy ProSavin, which was tested in a Phase I/II open-label study in 15 patients and demonstrated statistically significant improvements in motor behavior. AXO-Lenti-PD has been engineered to increase dopamine production compared with ProSavin, which could lead to a more efficacious gene therapy product.
To read the entire report Please click on the pdf File Below: