Oxford Biomedica (LON:OXB) has today announced a significant agreement with Novartis for the ongoing commercial and clinical supply of lentiviral vectors used to produce CTL019, confirming its status as a major component of Novartis’ future CAR-T plans. The agreement is an extension of the October 2014 collaboration and comes in anticipation of the launch of CTL019 in H217. In deal terms, OXB will receive a $10m upfront payment and could potentially receive $100m+ from Novartis over the next three years. Our forecasts and valuation are under review (in our last published outlook we noted peak royalties of £12.4m). We await the outcome of CTL019’s FDA advisory committee meeting (12 July) to shed more light on its path to approval and likely commercial success.
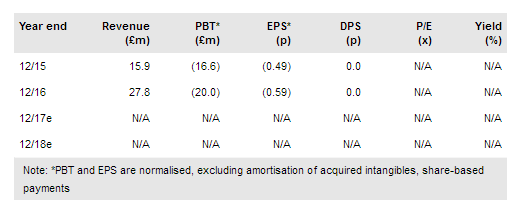
OXB’s agreement with Novartis for the commercial and clinical supply of lentiviral vectors used to generate CTL019 (tisagenlecleucel) and other undisclosed CAR-T products builds on the original contract in October 2014 (the supply agreement announced today is for three years, extendable to five years). OXB is the sole supplier of the lentiviral vector for the CTL019 clinical study, and the extension of the deal comes ahead of the anticipated clinical launch of CTL019 (FDA approval expected in H2 2017 in paediatric-ALL). OXB will additionally receive undisclosed royalties on potential future sales of Novartis’s CAR-T products including CTL019. In a separate press release last week, OXB announced the refinancing of the Oberland capital facility with a $55m debt facility from Oaktree Capital Management – Strategic Credit Strategy (“Oaktree”). Notably improved deal terms (potential cost of loan 11.5% compared to 15% for the previous Oberland facility) will aid OXB as it looks to become profitable in the near term. The loan is repayable no later than 29 June 2020 although it may be repaid, at the group’s discretion, at any time subject to early prepayment fees and an exit fee.
To read the entire report Please click on the pdf File Below: