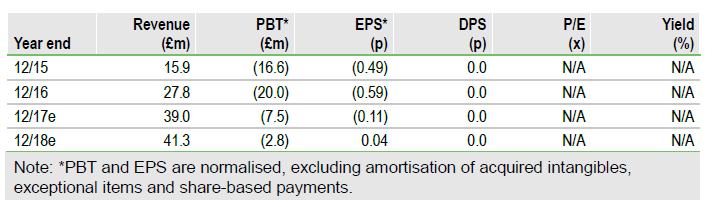
We expect Oxford Biomedica’s (LON:OXB) (OXB’s) strategic vision to come to further fruition through 2017/18 with both the potential approval of Novartis’s CTL019 in the US by year end and the possible spin-out/out-licensing of its priority development pipeline assets (OXB-102, OXB-202, and OXB-302). Full year 2016 results revealed robust growth in partnering revenues and 2017 will benefit from lower R&D expenses; we forecast a narrowing of EBITDA loss for the year. The net £17.5m equity fundraising and the Oberland debt facility has extended the current cash runway to 2019, aided by the reduction in near-term R&D; further funding and value may arise from additional manufacturing or IP licensing deals. Our revised valuation for OXB is £208.5m (6.75p/share).
CTL019 next steps: Potential for US launch in 2017
The global registration trial (ELIANA) of CTL019 in paediatric r/r B-cell acute lymphoblastic leukaemia (B-ALL) demonstrated overwhelming efficacy; 82% of patients treated achieved a complete remission or complete remission with incomplete blood count recovery. FDA have accepted Novartis’ Biologics License Application (BLA) and granted priority review for CTL019. We assume launch in 2017 (US) and Europe (2018) to directly benefit OXB, given its lentiviral technology remains vital to the manufacture of CTL019. We currently assume that a substantial portion of the $76m receivable from Novartis under the October 2014 contract has been delivered by mid-2017.
To read the entire report Please click on the pdf File Below