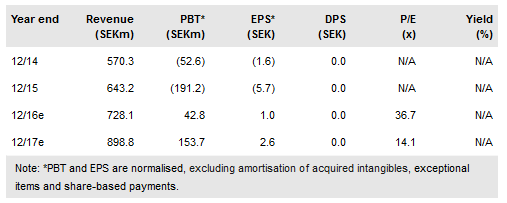
The court ruling upholding the validity of the Orexo AB (ST:ORX) ’996 Zubsolv patent precludes Actavis (NYSE:AGN) from launching a generic before September 2019. Orexo has 30 days to decide whether to appeal the court decision that a second patent (’330 with 2032 expiry) is invalid. A worst-case scenario allowing imminent launch of Actavis generic(s) has been averted. Zubsolv’s IP portfolio includes two additional patents (’900 and ’421) that extend to 2032, which coupled with the prospect of an appeal on ’330, means that significant hurdles remain ahead of Actavis generic launch.
Multi-layer IP strategy for Zubsolv
Actavis is the only company to file ANDAs for generic Zubsolv to date. The recent ruling relates to the June 2016 court hearing concerning the first ANDA (filed 2014 covering Zubsolv 1.4mg and 5.7mg); the court decision upheld the validity of the 8,454,996 US patent but found 8,940,330 invalid. Since the first ANDA filing, the FDA has approved four further Zubsolv dose strengths and two new patents have been issued (9,259,421 and 9,439,900). Actavis’s second ANDA was filed in 2015; Orexo responded by initiating two additional infringement suits. The first suit has an October 2017 trial date; the second (related to ’421) is pending a court date.
To read the entire report Please click on the pdf File Below